Translating biological insight into clinical investigation
Insights into retinal signaling and circadian network regulation have informed the development of Celeste as a therapeutic platform.
Early clinical studies demonstrated signals of improvement across several Parkinson’s disease symptom domains. These findings informed the design of the fully enrolled Phase 3 pivotal trial evaluating Celeste in Parkinson’s disease.
From Signal to
Pivotal Evidence
Celeste has progressed through early feasibility and Phase 2 clinical studies, culminating in a fully enrolled Phase 3 pivotal trial evaluating its potential to address non-motor symptoms in Parkinson’s disease.
Celeste has demonstrated clinically meaningful signals across motor and non-motor domains in earlier clinical studies. These findings informed the design of a fully enrolled Phase 3 pivotal trial evaluating Celeste in Parkinson’s disease.
Expected topline results are anticipated in 2026.
| Stage | Description |
|---|---|
| Early Feasibility | Initial studies explored the safety and biological effects of targeted photo-neuromodulation in Parkinson’s disease. |
| Phase 2 Study | Randomized, controlled study demonstrating signals of improvement across motor and non-motor symptom domains. |
| Pivotal Trial | Fully enrolled Phase 3 study evaluating safety and effectiveness in 350 participants across U.S. clinical sites. |
| Data Readout | Topline results anticipated in 2026. |
| Regulatory Pathway | FDA Breakthrough Device designation supporting regulatory engagement. |
The pivotal trial is fully enrolled and data are on the horizon.
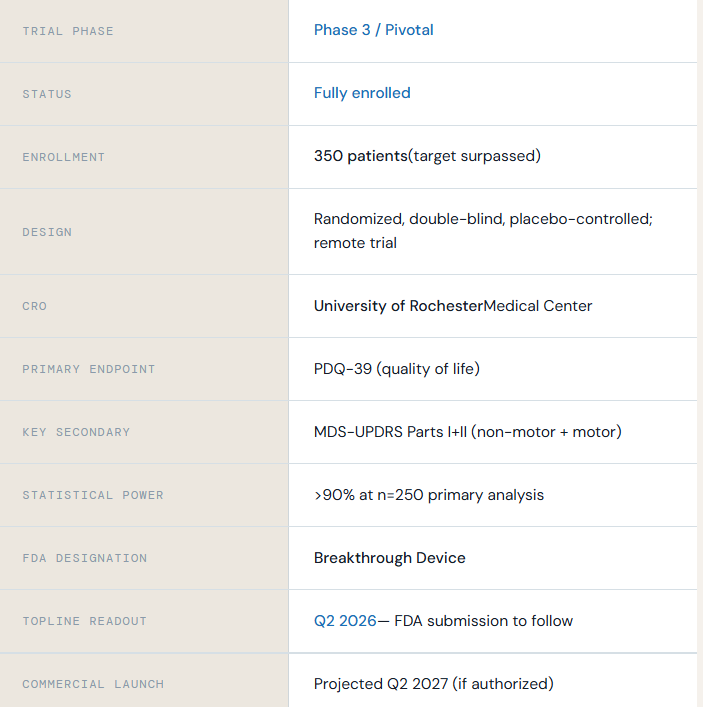
The Phase 3 clinical trial is designed to evaluate the safety and effectiveness of Celeste in people living with Parkinson’s disease.
The study builds on earlier clinical findings and was developed in consultation with regulatory authorities. It follows the same core therapeutic approach evaluated in earlier studies.
350 participants were enrolled across multiple U.S. centers.
-
Study phase
-
Enrollment
-
Design
-
Duration
-
Primary endpoint
-
FDA Breakthrough Device
Phase 2 established signals that informed the pivotal trial
Earlier clinical studies evaluating Celeste demonstrated signals of improvement across several Parkinson’s disease symptom domains. These findings informed the design of the Phase 3 pivotal trial now underway.
Motor symptoms
Clinically meaningful improvement in motor function relative to baseline.
Quality of life
Improvement in patient-reported quality-of-life measures.
Sleep and fatigue
Signals suggesting improvement in sleep-related outcomes.

Efficacy
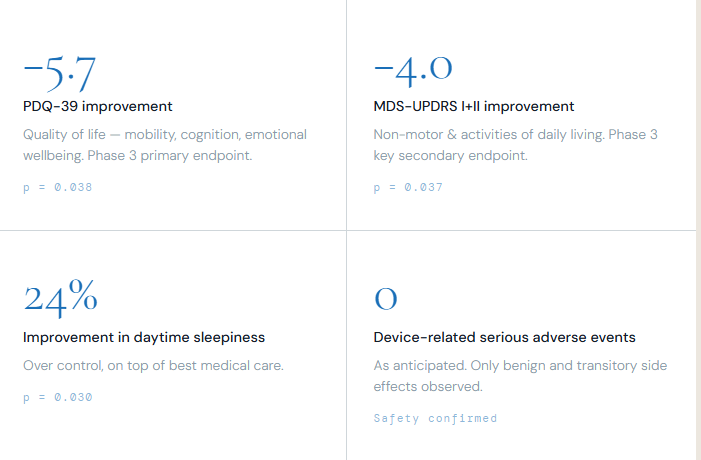
We saw clinically meaningful improvements in combined both motor and non-motor function and quality of life. Patients enjoyed statistically significant improvements in tremor, sleep, fatigue, cognition, depression, anxiety, apathy and urinary function. Preliminary trial results can be viewed here.
Safety
As anticipated, no device-related serious adverse events were reported in the trial. Patients experienced only benign and transitory side effects.
Supporting Research
Previous short-term studies have explored the potential effects
of targeted light exposure on neural signaling pathways associated with Parkinson’s disease.
These findings helped inform the design of Celeste and its ongoing clinical evaluation.
Building a scalable photo-neuromodulation platform
Parkinson’s disease is the proof point. The platform extends to further neurodegenerative conditions where non-motor burden is significant and entirely unaddressed.

Randomized Phase 2 Trial Completed
Published data established safety parameters and informed Phase 3 design.
Phase 3 Pivotal Trial Fully Enrolled
Topline Data Anticipated 2026
Readout intended to inform regulatory pathway under predefined primary and key secondary endpoints.
Potential Regulatory Submission
Platform Potential
Photo-neuromodulation targeting retinal signaling pathways may have relevance across multiple neurodegenerative conditions characterized by circadian and neural network disruption.
Celeste is currently being evaluated in Parkinson’s disease.
of Care
